Course:FNH200/Lessons/Lesson 12/Page 12.2
12.2 Dose Response and Risk Analysis
"All substances are poisons; there is none which is not a poison. The right dose differentiates a poison and a remedy" is a statement attributed to the 16th-century Swiss physician Paracelsus, leading to the basis of toxicology as we now know it. (Winter and Francis, 1997. Food Technol., 51:85).
In other words, every chemical and, more specifically in the context of this lesson, every toxicant has some set of exposure conditions in which it is toxic. Conversely, every chemical has some set of exposure conditions in which it is nontoxic.
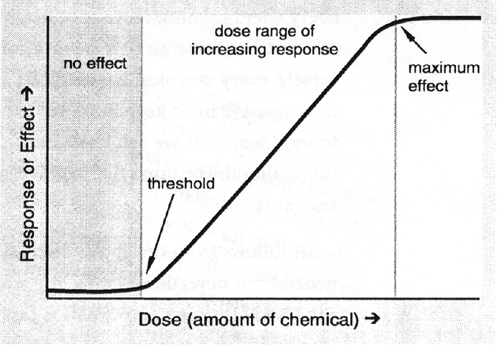
This can be described by the generalized dose-response curve shown in Figure 12.1.
The dose at which a substance begins to have an undesirable effect, that is, the upper limit of its "no effect" dose, is its threshold. This value is unique for each substance.
Furthermore, the slope or steepness of the increasing dose-increasing effect portion of the curve is also characteristic of the particular substance. A chemical with a very steep dose-response curve offers very little flexibility in trying to avoid a harmful exposure. A one- or two-fold difference in the amount of chemical consumed might be the difference between no-effect and serious consequences.
Both toxicity (capacity for injury as indicated by the threshold and dose-response curve) and hazard (toxicity under the conditions of exposure) are important in evaluating risks of toxicants in food. When considering toxicants we are in fact identifying a risk associated with eating. However, toxicants are only one such risk. To counter the risks are all the benefits associated with food consumption, such as provision of nutrients, pleasurable sensations, and so forth. We must weigh the risks and benefits associated with any activity, including eating food and make our decisions accordingly.
Review what we learned in Lesson 4 regarding the concepts of ADI, PDI, the "no-effect level" and the safety factor, as applied to the assessment of food additives. Similar considerations are involved in the assessment of the relative danger or safety of toxicants in our food supply.
The use of a safety factor is necessitated by our inability to account for all possible differences between human and animal, and among different humans, with absolute certainty. It should be pointed out, that proper toxicological evaluation of a chemical, attempts are made to take into consideration factors such as species differences and the influence of age, gender and environment of the animals used.