Course:FNH200/Lessons/Lesson 12
Toxicants in Food and Foodborne Disease
12.0 Overview
Foodborne disease affects a substantial portion of the Canadian population each year. The causes of food borne disease (etiolgy) includes agents with microbiological, parasitic, plant, animal and chemical origins.
In this lesson, we explore some examples of toxicants in foods, including naturally occurring constituents, naturally occurring contaminants and environmental contaminants. In order to understand the significance of toxicant presence and the regulations pertaining to them, the concepts of risk/benefit analysis toxicity, hazard, and acceptable daily intakes are discussed.
We also examine the relevant statistics to determine the major causes of foodborne disease and to gain an insight into factors that lead to foodborne disease outbreaks. We will also learn about safe food handling and preparation practices.
Objectives
After completing this lesson, you should be able to:
- define toxicity, hazard and risk, in the context of toxicants in our food supply
- explain the importance of considering the dose-response relationship
- outline the process for risk assessment in regard to food safety risks
- list and describe examples of naturally occurring (constituent or contaminant) and environmental toxicants in the food supply
- assess your risk of food intoxication by each of these toxicants and ways to minimize this risk
- explain the relative importance of various factors as contributors to the incidence of food borne disease in Canada;
- gain some insight into the major foodborne disease causing microorganims, in terms of conditions and foods implicated in outbreaks, and means of preventing their growth or toxin production in foods
- assess the potential risk of contracting food borne disease and describe safe food handling practices to minimize this risk at home
Optional Reading
- Murphy, P. A., Hendrich, S. and Landgre, C. 2006.Understanding Mycotoxins. IFT Scientific Summary
- http://www.ift.org/Knowledge-Center/Read-IFT-Publications/Science-Reports/Scientific-Status-Summaries/Editorial/Understanding-Mycotoxins.aspx
- Murphy, P. A., Hendrich, S., Landgre, C. and Bryant, C. 2006. Food Mycotoxins. An update. IFT Scientific Summary
- http://www.ift.org/~/media/Knowledge%20Center/Science%20Reports/Scientific%20Status%20Summaries/mycotoxins_0606.pdf
Recommended websites
- The Kidney Foundation of Canada B.C. branch: E. coli bacteria: What You need to Know
- Canadian Food Inspection Agency Food Facts. "Causes of Foodborne Illness". http://www.inspection.gc.ca/english/fssa/concen/causee.shtml
12.1 What are Toxicants?
Terms to remember |
|
In earlier lessons, we discussed the chemical nature of foods. The chemical constituents together are responsible for the structure, texture or consistency, colour, flavour, smell and nutritional value of a food. Food may also contain other chemical entities which are less useful to us or might even pose a health risk to us. These chemicals often have a very important role in the metabolism or function of the plant or animal tissues which make up our food. It must be remembered that while we consume various plants and animal products, their structure and composition is not primarily to serve as human food, but to exist as growing and living biological entities. Some potentially harmful chemicals in these plant and animal products are said to be environmental toxicants or even contaminants. They are not normally part of the chemical mix of food for humans, but by various means have found their way into our food supply.
In order to appreciate better the role of toxicants in the safety of our food supply, it is necessary to discuss a number of concepts and define some of the terms.
- Toxicant - a poison or a poisonous agent.
- The term toxicant is derived from the Latin toxicum (meaning "poison") and the Greek toxikon ("arrow poison"). The term toxic conveys the meaning that something is harmful, destructive or deadly.
- Poisons are chemicals that, in very small quantities, produce illness or death. Legally, a poison is defined as a chemical that has a lethal dose of 50 milligrams or less of chemical per kilogram of body weight.
- Fifty mg/kg is equal to approximately three-fourths of a teaspoon for an average adult and about one-eighth of a teaspoon for an average two-year-old child (Reference: M.A. Ottoboni. 1997.The Dose Makes the Poison: A Plain Language Guide to Toxicology. 2nd edition. NewYork: Wiley)
- When we refer to toxicants in food, we are generally referring to substances responsible for a whole spectrum of possible results, ranging from relatively minor discomfort or sickness to poisoning that can lead to death.
- Toxicity - the intrinsic or inherent capacity of a substance to damage a biological system (produce injury) when tested by itself.
- Substances vary in their toxicity, as reflected in their dose-response curves.
- A toxicant can have an effect on several different functions within an individual. The individuals can vary among themselves with regard to the sensitivity of their different functions.
- Hazard - A thing or action that can cause adverse health effects in animals (including fish), plants or humans. The capacity to produce injury under the circumstances of exposure.
- Hazard is a much more complex concept than toxicity because it includes a consideration of conditions of use; in other words, two components are involved in assessing a hazard:
- 1. the inherent capacity to cause harm (toxicity), and
- 2. the ease or probability of contact between the substance and the target object.
- These two components together describe the chance or probability that a substance will do harm. Dr. Ottoboni (cited below) provides two examples to make this clear.
- Hazard is a much more complex concept than toxicity because it includes a consideration of conditions of use; in other words, two components are involved in assessing a hazard:
"An extremely toxic chemical, such as strychnine, when sealed in an unopenable vial, can be handled freely by people with no chance that a poisoning will occur. Its toxicity has not changed, but it presents no hazard because no contact can be established between the chemical and people. Conversely, a chemical that is not highly toxic, such as boric acid, can be very hazardous when used in a manner that makes it readily available for accidental ingestion." Dr. Ottoboni
Risk - a function of the probability of an adverse health effect and the magnitude of that effect, consequential to a hazard; the likelihood of the occurrence and the magnitude of the consequences of an adverse event.
12.2 Dose Response and Risk Analysis
"All substances are poisons; there is none which is not a poison. The right dose differentiates a poison and a remedy" is a statement attributed to the 16th-century Swiss physician Paracelsus, leading to the basis of toxicology as we now know it. (Winter and Francis, 1997. Food Technol., 51:85).
In other words, every chemical and, more specifically in the context of this lesson, every toxicant has some set of exposure conditions in which it is toxic. Conversely, every chemical has some set of exposure conditions in which it is nontoxic.
This can be described by the generalized dose-response curve shown in Figure 12.1.
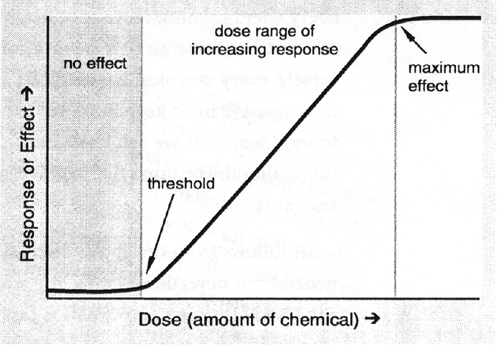
The dose at which a substance begins to have an undesirable effect, that is, the upper limit of its "no effect" dose, is its threshold. This value is unique for each substance.
Furthermore, the slope or steepness of the increasing dose-increasing effect portion of the curve is also characteristic of the particular substance. A chemical with a very steep dose-response curve offers very little flexibility in trying to avoid a harmful exposure. A one- or two-fold difference in the amount of chemical consumed might be the difference between no-effect and serious consequences.
Both toxicity (capacity for injury as indicated by the threshold and dose-response curve) and hazard (toxicity under the conditions of exposure) are important in evaluating risks of toxicants in food. When considering toxicants we are in fact identifying a risk associated with eating. However, toxicants are only one such risk. To counter the risks are all the benefits associated with food consumption, such as provision of nutrients, pleasurable sensations, and so forth. We must weigh the risks and benefits associated with any activity, including eating food and make our decisions accordingly.
Review what we learned in Lesson 4 regarding the concepts of ADI, PDI, the "no-effect level" and the safety factor, as applied to the assessment of food additives. Similar considerations are involved in the assessment of the relative danger or safety of toxicants in our food supply.
The use of a safety factor is necessitated by our inability to account for all possible differences between human and animal, and among different humans, with absolute certainty. It should be pointed out, that proper toxicological evaluation of a chemical, attempts are made to take into consideration factors such as species differences and the influence of age, gender and environment of the animals used.
12.3 Types of Toxicants
Food toxicants can be classified in a variety of ways. The scheme in Table 12.1 is based on origin or occurrence of the toxicants, namely as naturally occurring toxicants (either constituents or contaminants), or environmental toxicants.
Table 12.1.Classification of some possible food toxicants
| Naturally Occurring Toxicants:
Constituents |
Naturally Occurring Toxicants:
Contaminants |
Environmental Toxicants |
|---|---|---|
Cholinesterase inhibitors:
Cyanogenic glycosides:
Glucosinates:
Protease inhibitors:
Nitrites:
Allergens:
|
Mycotoxins:
Examples: aflatoxin in mouldy peanuts, vomitoxin in wheat Bacterial toxins:
Seafood Toxins:
|
Food packaging residues:
Pesticide residues:
Example: malathion residues on fruit Heavy metals:
Examples: lead in vegetables; mercury in large ocean fish (eg. swordfish) Animal drugs:
Examples: penicillin in milk Radioisotopes:
|
12.4 Natural Constituents and Natural Contaminants
What is the basis for distinction between Natural Constituents and Natural Contaminants?
Constituents are chemical entities that are part of the normal composition of a food material - they are not the result of some external organism or activity. Contaminants, on the other hand, are present because of the presence of moulds or bacteria, or because the plant or animal was grown in a condition which permitted the toxicant to become part of the food.
The distinction between natural and environmental toxicants is a little bit less clear cut, but may be generally determined by their origin. For example, we might be able to prevent the growth of moulds, but moulds are nevertheless naturally present in the environment. Pesticides, however, are introduced into the environment by us and while not intended to become part of the food, some do to a certain extent. Mercury and lead can enter the food supply because of heavy natural deposits in the soil, but in fact are found in food predominantly because we use these metals in a wide variety of ways.
It is difficult to be definitive about the relative importance of these different food toxicants to human health. Much of what has been written on this subject can be summarized by the information in Table 12.2.

This table attempts to illustrate the perceived importance of hazards in food. It is interesting to note that what is perceived by popular opinion to be the greatest hazard is in fact considered to be the least important by regulatory agencies, and vice-versa! Regulatory agencies base their ratings on a much broader information base (including statistical or epidemiological evidence) than does the general public.
An extensive discussion of each of the toxicants identified in Table 12.1 is not possible. Several will be discussed in some detail below, followed by a brief comment on the remaining ones.
12.5 Examples of Natural Constituents as Toxicants
Glycoalkaloid-Cholinesterase Inhibitors
- The name "cholinesterase inhibitors" refers to a variety of chemicals which are able to inhibit the activity of the enzyme cholinesterase. This enzyme is found in nerve tissues and plays an important role in the transmission of nerve impulses. When its function is inhibited, nerve function is affected.
- Solanine is an example of such inhibitors. Solanine can is found in potatoes, normally at levels of 2-13 mg/100 g fresh weight,
- the bulk of market potatoes probably contain only 3-6 mg/100 g.
- Levels as high as 80-100 mg/100 g, have been reported particularly if the potatoes have undergone greening (reaction of potato tubers on exposure to sunlight).
- Solanine is insoluble in water and is not lost or destroyed when potatoes are cooked. Because it is found primarily in the skin of potatoes, the peeling of potatoes reduces the concentration markedly.
Clinical symptoms of solanine poisoning: gastrointestinal disturbances and certain neurological disorders. Solanine poisoning can result in death. However, ingested solanine is less likely to cause toxic symptoms than an injected dose because it is not readily absorbed and is fairly rapidly excreted by way of the feces and the urine. Humans display drowsiness, increased sensory sensitivity, and difficulty in breathing after an oral dose of 100 mg (approximately 2-8 mg/kg body weight). Higher doses may cause vomiting and diarrhea.
| Want to learn more? |
|
- Although the chemical solanine itself is quite a potent toxicant, the hazard of solanine poisoning by consumption of potatoes is quite low.
- Potato growing and handling practices minimize the opportunity for greening, and hence minimize the production of abnormally high levels of solanine.
- Seed potatoes which are genetically low in solanine content are used, and during the growing season the potato plants are hilled, thereby decreasing exposure of the potato to light.
- After harvesting, potatoes are put into storage facilities which minimize exposure to light.
- Consumers who ignore the hazards of consuming potatoes that have turned green during improper storage at home would face a high risk of poisoning by solanine!
| Want to learn more? |
|
Cyanogenic Glycosides
- Cyanogenic glycosides yield hydrogen cyanide (HCN) upon treatment with acid or particular hydrolytic enzymes.
- They are found widely in higher plants, and also occur in ferns, moths and insects. Among the plants used as food which contain one or more of these toxicants are: cassava, sweet potato, yam, maize, bamboo, sugar cane, peas, lima beans, almonds, lime, apple, pear, cherry, apricot and plum.
- Cyanide is very rapidly absorbed from the gastrointestinal tract and produces recognizable symptoms at both fatal and non-fatal levels.
With fatal doses of HCN, death results from the general anoxic condition caused by the inhibition of cytochrome oxidase, with which the HCN complexes. Cytochrome oxidase is an important component of the oxidative phosphorylation cycle which occurs in the mitochondria of cells. Inhibition of the enzyme system causes the death of the cells.
Since the HCN binds reversibly to the cytochrome oxidase, non-fatal doses permit recovery by means of respiratory exchange and metabolic detoxification processes.
For humans, the minimum lethal dose of HCN taken orally has been estimated to be between 0.5-3.5 mg/kg of body weight. It is obvious from the data in Table 12.3 that consumption of 100 g of bitter almonds by a 70 kg man could produce a dose of 3.57 mg/kg of body weight, a fatal dose even for the least sensitive individual.
- Amygdalin is an example of a cyanogenic glycoside. It is found in bitter almonds and a number of fruit pits.
- Amygdalin, having a carbohydrate component ("glycoside") and hydrogen cyanide is hydrolyzed to form HCN, glucose and benzaldehyde ( Figure 12.2).
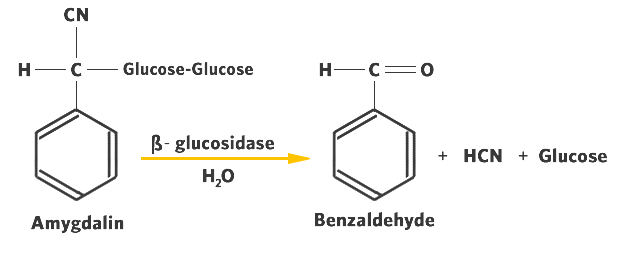
The enzymes responsible for the hydrolysis of cyanogenic glycosides are generically called ß-glycosidases. They are highly specific for the ß-glycosidic linkage that is characteristic of the cyanogenic glycosides.
These plant enzymes differ from the a-glycosidases (amylases) of the mammalian digestive tract that hydrolyze only a-glycosidic bonds such as those found in starch.
- Hydrolysis of cyanogenic glycosides can occur during the cutting, crushing, bruising or maceration of tissue prior to consumption, or can be initiated upon the maceration of the tissue during eating.
- Cyanide poisoning can therefore occur when enough of a plant material that is rich in cyanogenic glycosides and enzymes is consumed, or when such a material has been prepared with insufficient care to remove the HCN accumulated during preparation.
The scientific literature records numerous incidents of accidental poisonings by bitter almonds, cassava, and lima beans. Table 12.3 gives some indication of the amount of cyanide that may be produced from various plant foodstuffs.
Table 12.3 Data showing the potential yield of cyanide in some plant foodstuffs. Adapted from: Liener, I.E. (ed.). 1980. Toxic constituents of plant foodstuffs. New York: Academic Press.
| Plant | HCN Yield
(mg/100 g material) |
|---|---|
| Bitter almonds | 250 |
| Bitter cassava root | 53 |
| Bitter cassava root cortex (dried) | 245 |
| Lima bean, American white | 10 |
| Lima bean, Java coloured | 312 |
- In parts of the world where some of these cyanide producing plants are used extensively for food, means of preparation have been developed to remove or hydrolyze the glycosides and to destroy the ß-glucosidase that is present. While these steps minimize the hazard, they do not completely eliminate it.
It is important, however, that while the cyanide-producing capacity of a plant is important in determining its toxicity, there are other factors which must also be considered. These factors include the size and kind of subject, the speed of ingestion, the type of food ingested simultaneously with the cyanogen, the presence of active degradative enzymes both in the plant and in the subject's digestive tract, and the subject's ability to detoxify the HCN.
The hazard associated with the presence of cyanogenic glycosides in food for those individuals who consume large quantities of the affected plant foodstuff, is much more significant than those who accidentally eat the occasional apple seed or apricot kernel.
Other Natural Toxicants
There are a number of other natural toxicants listed in Table 12.1 for which only a very brief comment will be made in this course.
Protease inhibitors
- Proteinaceous compounds found in many of the legume species.
- have the ability to complex to, and thereby interfere with, certain proteolytic enzymes.
- Proteins need to be hydrolyzed into their constituent amino acids by digestive enzymes such as trypsin and chymotrypsin. If these enzymes are rendered inactive by complexing inhibitors, the body cannot fully hydrolyze the proteins, thereby creating the possibility of amino acid deficiencies.
Nitrates
- Widely found constituents of plant materials, especially green leafy plants.
- Nitrates themselves are not very toxic; however, bacteria can reduce them to nitrites.
- A primary concern about nitrites is their ability to interact chemically with hemoglobin, interfering with the blood's ability to transport the required oxygen to the body's cells. By a rather complex series of reactions, not only bacteria but also metabolic pathways within the digestive system of humans can utilize nitrate/nitrite as a precursor for the formation of nitrosamines, potent carcinogens.
Allergens
- Have the ability to induce allergic reactions in sensitive individuals. Foods that are most frequently reported to cause allergic responses are: cereals such as wheat, rye and rice; legumes such as peas, peanuts and soybeans; tree nuts; milk; eggs; and seafoods such as shrimp, crab and lobster.
- The case of allergens provides a good example of the need to consider individual (genetic) differences in assessing risk and hazards. For most of us, consumption of the foods listed above does not pose any significant risk. However, for someone who is severely allergic to a particular food or food component, accidental ingestion may lead to a life-or-death situation!
12.6 Examples of Natural Contaminant as Toxicants
Mycotoxins
Mycotoxins are substances produced by moulds, which may be toxic. Mycotoxins may occur by direct contamination (due to mould growth on the food) or by indirect contamination (by using a food ingredient that was contaminated). Mycotoxins can be highly toxic to the body, some have been known to cause cancer in animal tests, others are mutagenic and able to cause mutation, and others are teratogenic and capable of causing deformities in embryos. Mycotoxins are invisible to the eye, are often unchanged by heat and can potentially spread throughout a product. As a result, any sign of visible mould could indicate that the product contains mycotoxins and should be thrown away.
| Want to learn more? |
|
Of the many known mycotoxins (aflatoxins, sterigmatocystin, ochratoxin A, citrinin and patulin), the aflatoxins are of greatest concern because they are potent liver toxins in all animals in which they have been tested and carcinogens in some species. The different aflatoxins (B1, B2, G1, G2, M1and M2) may be found in various commodities including peanuts, corn, wheat, rice, cottonseed, copra, nuts, milk, eggs, and cheese.
One additional comment needs to be made in connection with the regulation of mycotoxins. Aflatoxins are not directly regulated in The Food and Drugs Act. In Canada, this has caused some problems in the past. A challenge of the contention that aflatoxin was injurious to human health caused a court to dismiss a charge of selling contaminated peanut butter that contained greater than 20 ppb of aflatoxin. To get around that problem, the act and regulations were changed to state that peanut butter would be considered contaminated, and therefore not fit for sale, if it contained aflatoxins in excess of 15 ppb.
Generally, any food which contains mould should be thrown out however because the risk of mycotoxins is fairly low in some instances, the benefit of saving the product may outweigh the potential risks involved.
The following should be considered when we encounter a mouldy food:
- Was the food refrigerated? Did the mould grow on the product while it was refrigerated? If it has, it is likely that the product does not contain aflatoxins (these are not normally produced under refrigerated conditions).
- The amount of mould on the product should also be considered, for example, if there is a tiny green mould colony on a large block of cheese, the benefits of cutting that portion off (about 2.5 cm around and beneath the cheese) are much greater than the risks involved, especially relative to the benefits of trimming extensive mould growth on a tiny piece of cheese.
- The type of food the mould is growing on should also be considered; “trimming” mould should not be done on soft, semi-solid, or liquid foods, or foods likes jams/jellies and baked products. In these products, the ability for the mould toxins to spread (via the mould mycelia) are much greater. In these cases, the risks outweigh the benefits of saving the product.
- To ensure that mould growth does not occur at home, consumers can try to prevent mould growth by minimizing contact of food with air as well as package (wrap) and refrigerate products appropriately.
| Critical Thinking |
|
Bacterial Toxins
The two most important bacterial toxins are those produced by Clostridium botulinum and Staphylococcus aureus, which are ubiquitous microorganisms in our environment. Food poisoning caused by these two types of bacteria can be considered as due to food intoxications, because it is the chemical toxin produced by the bacteria, as opposed to the bacteria themselves (in the case of foodborne infections), that produces the toxicity. The toxins can produce illness even if the bacteria producing them have been killed. We will explore these in further detail later in this lesson (under "foodborne diseases").
Check out the recent Health Hazard Alerts regarding possible Clostridium botulinum contamination.
http://www.inspection.gc.ca/about-the-cfia/newsroom/food-recall-warnings/complete-listing/2019-03-15/eng/1552702897100/1552702899415
Seafood Toxins
Most of the natural toxicants that we encounter in our food supply are in products of plant origin. Animal tissues are much less frequently contaminated with natural toxicants. Part of the reason for this might be the fact that animals are very similar in their make-up and metabolism to humans. Animals would react similarly to toxins in their environment as humans and consequently we are not confronted with animal tissues that are from diseased or poisoned animals. For this reason, while it is a somewhat broad generalization, most natural toxicants in foods categorized as animal origin are found in seafoods rather than other animal products, and in fact, in many of these cases, the source of the toxicants originate from marine algae or through the action of microorganisms.
There are many types of seafood toxins; here we will review some of the most common ones: Histamine, Saxitoxin, Domoic acid, and Tetrodotoxin.
Histamine
Poisoning is known to result from eating scombroid fishes, i.e. fish of the Scombrida and Scomberesocidae families, including tuna, bonito and mackerel. The poisoning results in nausea, vomiting, facial flushing, headache, epigastric pain, thirst, itching of the skin and hives. The effects usually subside within 12 hours but in some severe cases death has resulted.
Scombroid poisoning is due to an allergic-type reaction to high levels of histamine or a histamine-like substance. Histidine, a naturally occurring amino acid which is particularly high in scombroid fish, is converted to histamine by bacterial action on the dead flesh of the fish. Histamine has strong vaso-active properties and in sufficient quantities will cause blood pressure changes resulting in symptoms as indicated above. It should be noted that toxic amounts of histamine may be formed before the fish starts to smell or taste bad ("spoil"). Histamine formation is dependent on the holding time and temperature of the fish during transportation and storage.
It is also important to note that histamine and other vaso-active amines may be found in many fermented foods such as cheese, wine and pickled herring.
Saxitoxin
Saxitoxin and a group of 18-24 marine biotoxins derived from saxitoxin are thought to be responsible for the condition known as paralytic shellfish poisoning (PSP). This condition is the result of consuming shellfish, such as mussels, oysters, and clams, containing the toxin. The toxins can also accumulate in the liver of crustaceans such as crabs and lobsters.
The toxin is ineffective against the shellfish, which are able to absorb the toxin, concentrate it in their tissues and pass it onto the species feeding on them (in this case, humans).
Symptoms of saxitoxin include: tingling in the mouth, lips and finger tips. Sight and hearing are modified in much the same way as caused by alcohol intoxication. Speech becomes incoherent.
Saxitoxin is a contaminant which the shellfish consume as part of their occasional food supply. The shellfish feed on a variety of microscopic plankton among which are certain toxic dinoflagellates. At certain times of the year, under certain conditions, these dinoflagellates increase greatly in number and concentration in the oceans. This is often referred to as a bloom or "red tide."

During periods of bloom or red tide, the shellfish are exposed to and concentrate large amounts of saxitoxin. Saxitoxin is extremely stable and takes a long time to be flushed from the tissues of the shellfish. It is for this reason that during red tide season and for a considerable time afterwards, harvesting of shellfish is prohibited.
Saxitoxin is considered one of the potent toxins, the minimum lethal dose being about 1.0-4.0 mg/kg of body weight. However, because of the variability in the concentration of toxin in the shellfish, it is very difficult to indicate a maximum amount of shellfish tissue that can be consumed. This is obviously a situation which is not at all hazardous to anyone who rarely harvests and consumes shellfish. However the hazard increases significantly, for those who are uninformed and are fond of shellfish, particularly those harvested in areas known to be subject to red tide situations.
Domoic Acid
Domoic acid is (an analog of the amino acid glutamic acid), is found in some marine algae (dinoflagellate Nitschia), which can accumulate in filter feeding shellfish such as clams, mussels, scallops and oysters. Consumption of shellfish with this naturally occurring marine biotoxin can lead to amnesic shellfish poisoning (ASP).
Domoic acid, a neurotoxin, is capable of causing lesions in the central nervous system. It was first discovered in 1987 in Canada, when an outbreak occurred in individuals consuming mussels in Prince Edward Island. Of 107 individuals who were affected, four died. Most experienced gastroenteritis, and many older persons, or others with underlying chronic illnesses, developed neurological symptoms including memory loss. In this case, enough toxin was absorbed through the gastrointestinal system to cause the effects. The most severely affected cases still had significant memory loss five years after the incident.
Routine testing for the toxin is conducted, and areas in which the toxin occurs in the shellfish are closed to harvesting. Thus, the risk of amnesic shellfish poisoning is high only for individuals who choose to ignore signs indicating closure of areas to shellfish harvesting.
If you regularly harvest your own shell fish for consumption, remember to check for both PSP (and other marine toxins) and Sanitary Contamination Closures at this Fisheries and Oceans Canadawebsite - Shellfish Contamination Closures - Pacific Region (Links to an external site.)
http://www.pac.dfo-mpo.gc.ca/fm-gp/contamination/biotox/index-eng.html (Links to an external site.)
Tetrodotoxin
This toxin is the chemical of interest in puffer-fish or fugu poisoning. In Asian countries this hazard has been known for thousands of years. Although pufferfish have been known to be poisonous, many deaths still occur from eating these fish. Tetrodotoxin is found mainly in the ovaries, liver, intestine, skin and spawn of the various species of pufferfish. Although tetrodotoxin (TTX) was discovered in these fish, it is thought to be synthesized by a bacterial species such as Pseudoalteromonas tetraodonis associated with the puffer fish.
The meat of these fish species is considered a delicacy by the Japanese and Chinese, and the choice edible species are those that are most poisonous. The amount of poison in the fish is lowest in the summer months and increases during winter, with a peak just before spawning, in the spring.
Symptoms of pufferfish poisoning usually begin with a tingling sensation of the fingers, toes, lips and tongue a few minutes after eating the fish. Nausea, vomiting, diarrhea and epigastric pain appears in some cases. As the poisoning progresses, reflexes of the pupil and cornea are lost and the patient increasingly experiences paralysis and respiratory distress. If the dose is sufficiently large, death will result, caused by respiratory paralysis. The mode of action of tetrodotoxin is considered to be essentially the same as saxitoxin. As with saxitoxin, a lethal dose for humans is thought to be about 1.0 - 4.0 mg/kg.
Preventive measures are almost entirely restricted to diligence during preparation of the fish. When gutting and cleaning the fish, care must be exercised so as not to contaminate the flesh with even the minutest amount of viscera, ovaries or spawn.
Pufferfish poisoning continues to be a problem in Japan, affecting 30 -100 persons per year, and primarily as a result of home preparation and consumption, not from commercial sources of the pufferfish. Again we have an example of an extremely toxic chemical but one whose hazard is quite variable. Lovers of fugu are at considerable risk, while those who do not consume this food are at absolutely no risk.
| Want to learn more? |
|
12.7 Examples of Environmental Toxicants
Table 12.1 lists a rather wide range of environmental toxicants, but this list is not comprehensive. They include:
- products of industrial activity (e.g. mercury, lead, dioxins, polychlorinated biphenyls, radioisotopes)
- products of agricultural activity (e.g. pesticide and herbicide residues, antibiotic residues)
- products of food processing (e.g. packaging residues)
- naturally occurring environmental toxicants (e.g. mercury, radioisotopes)
You may have heard about the tragic news in 2005 of the death of 27 children in Manila (the Philippines), after eating deep-fried caramelized cassava sold by vendors as recess snacks to the children. More than 100 other children, as well as one of the two vendors, were admitted to hospitals, suffering from severe stomach pain, vomiting and diarrhea. Although early reports suspected cyanide poisoning from improperly cooked cassava, subsequent laboratory tests on the cassava snack samples as well as analysis of the patients indicated that contamination by carbamate pesticide was the likely cause of the poisoning.
"Carbamate pesticide is commonly used in farms and households on Bohol, and may have already been in the environment." Source: The Vancouver Sun, March 10, 2005 and March 14, 2005.
Some environmental toxicants display their toxicity at concentrations many, many times greater than we are likely to find in food; others are toxic at concentrations not far removed from those found in foods. All of them are of concern because they inhabit the environment and hence can enter our food supply. But, more important, all of them are present in the environment because they have some property that makes them useful to us. As a consequence, we must examine all environmental toxicants from a risk/benefit perspective. The decision to continue to use them should be made because their benefit outweighs their risk. Their continuing presence in the environment may also happen to a time in the past when their benefit outweighed their risk, but that may no longer be the case; a current review of risk/benefit would be in order.
Due to time constraints, we will not explore the environmental toxicants in more detail in this course.
| Want to learn more? |
|
12.8 Foodborne Diseases
You may have suffered from a bout of foodborne disease or you may know of someone who has been a victim of foodborne disease. We often hear the term food poisoning but foodborne disease is the correct term.
According to the most recent estimate from the Public Health Agency of Canada the number of cases of Foodborne illness that occur in Canada is approximately 4 million cases each year.
A foodborne illness is caused by eating food that has been contaminated in some way. Contamination can be from different causes (etiology): microbiological, parasitic, plant, animal, and chemical.
- Microbiological agents are responsible for the majority of foodborne disease outbreaks. The specific microbiological agents are discussed in forthcoming sections of this lesson.
- Parasitic agents refer to parasites such as Trichinella spiralis, Anisakis simplex, tapeworm, and so forth, that are acquired from consumption of infested foods:
- Trichinella spiralis, associated with consumption of undercooked pork, contaminated water or produce.
- Anisakiasis caused by Anisakis simplex. Anisakiasis is associated with eating raw fish (sushi, sashimi, lomi lomi, ceviche, sunomono, Dutch green herring, marinated fish and cold-smoked fish) or undercooked fish.
- Foodborne diseases arising from consumption of plants included consumption of amanita mushrooms, taro leaves, green potatoes, and toxic algae in spirulina (an algae) protein supplement tablets.
- Illness arising from poisonous animals usually occurred as a result of consumption of shellfish containing paralytic toxins.
- Chemicals in contaminated foods that cause foodborne disease have included tin in canned foods arising from corrosion inside the metal can, residues of cleaning and sanitizing agents in food, products of rancid fats in foods, and excesses of some ingredients such as monosodium glutamate.
Here are some terms that you need to be familiar with:
- An incident refers to the occurrence of foodborne illness
- An outbreak is an incident in which two or more people experience a similar illness after ingestion of the same food and where epidemiological evidence implicates the food as the source of the illness.
- A single case is a person who has been ill following consumption of food considered to be contaminated on the basis of epidemiological evidence.
- An outbreak or single cases of unknown etiology refer to situations where epidemiological evidence implicates a common food source but where laboratory analysis fails to identify the etiological agent
Table 12.8 shows the number of cases reported in B.C. for certain types of microorganisms responsible for foodborne diseases.
| Table 12.8. Number of reported cases in British Columbia | ||||
|---|---|---|---|---|
| Microorganism | 2000 | 2004 | 2006 | 2014 |
| E. coli O157:H7 | 166 | 193 | 150 | 56 |
| C. botulinum | 4 | NA | NA | NA |
| Salmonella | 710 | 707 | 705 | 1167 |
| Listeria monocytogenes | 6 | 10 | 13 | 19 |
| Hepatitis A- Virus | 136 | 76 | 55 | 25 |
*All
Source: B.C. Centre for Disease Control. Annual Summary of reportable diseases. Enteric, Food and Waterborne diseases 2006. NA = Not available.
The numbers in Table 12.8 represent only the "tip of the iceberg" since epidemiological evidence suggests that only 1-4% of all foodborne disease outbreaks are actually reported and documented.
Some of the reasons why only some foodborne disease outbreaks are documented are:
- many people do not report to a physician when they suspect they are suffering from foodborne disease;
- actual cases of foodborne disease may be mistakenly diagnosed as having other sources (environmental, unknown sources);
- many people may attribute a mild case of foodborne illness as being due to the "24-hour flu," a "virus," "eating too much"'
- sometimes people report that they suspect they are suffering from foodborne illness but the physician does not report the case to local health units or, if reported, the case may not be sufficiently well researched and reported by health units to the provincial health authorities.
Most cases of foodborne illness are only recognized when at least two or more people become sick after eating a meal or food product in common ("outbreak"). Symptoms can arise hours or days following the consumption of the food. The CFIA reports that each year only about 10,000 cases of foodborne illness are reported from which approximately 30 people die. The cost of salmonellosis has been estimated at $850 million per year in hospitalization costs and lost income. If one were to add the costs of foodborne disease caused by other organisms and other agents, the total costs become staggering (in excess of 1.3 billion dollars per year in Canada). Similar foodborne disease trends are reported yearly in the United States, the European Economic Community, Australia and other developed countries.
12.8.1 Microorganisms and Foodborne Diseases
Table 12.9 shows examples of microorganisms that cause foodborne disease. Pathogenic microorganisms can take advantage of situations where safe food handling and storage practices are not followed.
Table 12.9. Examples of microorganisms responsible for foodborne diseases
| Microorganism responsible | Foodborne disease |
|---|---|
| E. coli O157H7 | E. coli poisoning |
| Toxin produced by Clostridium perfringens | Clostridium food poisoning |
| Toxin produced by Clostridium botulinum | Botulism |
| Salmonella | Salmonellosis |
| Listeria monocytogenes | Listeriosis |
| Staphylococcus aureus | Staph poisoning |
The following short descriptions of the microorganisms will provide you with an insight on the major foodborne disease-causing microorganisms, where they are found, conditions and foods implicated in outbreaks, and disease symptoms, as well as means of preventing their growth in foods.
For this course, you do not need to memorize all the details described here or in the readings. However, you should gain an understanding of how such detailed information about the different properties of pathogenic microorganisms can be used to provide guidelines for food handling practices or preservation techniques to ensure better food safety and to decrease the incidence of foodborne illness.
As you read through the descriptions, try also to differentiate between those microorganisms which are responsible for foodborne illness through food intoxications versus those that do so through foodborne infections.
- In the case of foodborne intoxications, toxic substances are produced in the foods as by-products of the microorganisms prior to consumption, and cause the symptoms of foodborne illness upon ingestion.
- Foodborne infections result from the ingestion of viable microorganisms in the food at the time of consumption; these pathogenic microorganisms multiply and grow in the host, causing the symptoms of foodborne illness.
Food Intoxications:
| Organism | Symptoms onset | Common foods | Prevention |
|---|---|---|---|
| Clostridium botulinum | Botulinal toxins are among the most toxic substances known. Estimated LD50 for humans is 1 ng/kg body weight.
Dizziness, double vision, difficult swallowing, respiratory or cardiac paralysis, death (12-36 hrs) |
Low acid foods in anaerobic conditions. Improperly home-canned, vacuum-packed, poorly refrigerated foods
Baked potatoes wrapped in foil Spores have been detected in vegetables such as carrots, potatoes and fruit (contamination with soil), in fresh and processed meats and in honey and corn syrup. |
Commercially sterile after processing must receive a "botulinum cook" (12 D)
Use sodium nitrite for cured meats Properly refrigerate vegetable/fruit juice. Honey, corn syrup and molasses must not be fed to infants < 1 year of age. Pasteurization of honey does not inactivate the spores. |
| Organism | Symptoms onset | Common foods | Prevention |
|---|---|---|---|
| Escherichia coli O157:H7(Shiga toxin) | Bloody diarrhea, kidney failure and hemolytic uremic syndrome (HUS) in sever cases (12-72 hrs) | Undercooked ground meat, raw milk, unpasteurized apple juice/cider, lettuce, spinach, alfalfa sprouts
Contaminated water supply (outbreak in Walkerton, Ontario) |
Practice good food sanitation and hand washing.
Properly handle and cook foods.E. coli is killed by normal cooking and pasteurization, as well as ionizing radiation, but is able to survive in acidic conditions (e.g. apple juice). It does not grow well at temperatures below 8°C. The Kidney Foundation of Canada B.C. branch: E. coli bacteria: What You need to Know |
Food Infections:
| Organism | Symptoms onset | Common foods | Prevention |
|---|---|---|---|
| Salmonella spp. | Nausea, fever, vomiting, abdominal cramps, diarrhea (6-48 hrs) | Raw meats and poultry, eggs (about 60% of chickens sold in the retail market are contaminated with Salmonella), milk, dairy products | Salmonella are easily killed by heat (normal cooking and pasteurization processes) and ionizing energy, but survive freezing and dehydration. Consequently, frozen and thawed poultry can be just as contaminated as fresh poultry. |
| Organism | Symptoms onset | Common foods | Prevention |
|---|---|---|---|
| Listeria monocytogenes | Flu-like symptoms, meningitis, septicemia, or pneumonia. Birth defects, still birth (1 day- 3 weeks) | Raw milk, dairy products, vegetables, fish and meat products | The bacteria is capable of growing slowly on foods during storage in the refrigerator, and appears to be fairly tolerant to salt. It is easily killed by proper cooking and pasteurization techniques. Refrigerate raw materials and high-risk foods below 4°C. |
Viruses
Viruses are much smaller than bacteria and require a "living host" (human, animal) in which to grow and reproduce. Viruses do not multiply in food. Viruses are usually transferred from one food to another, from a food worker to a food, or from a contaminated water supply to a food.
| Organism | Symptoms onset | Common foods | Prevention |
|---|---|---|---|
| Hepatitis A | Fever, nausea, vomiting, abdominal pain, fatigue, swelling of the liver, jaundice (10-50 days) | Foods prepared with human contact; contaminated water | Wash hands and practice good personal hygiene, avoid raw seafood |
| Organism | Symptoms onset | Common foods | Prevention |
|---|---|---|---|
| Noravirus | Vomiting, diarrhea, headache, fever (24-48 hrs) | Sewage, contaminated water, contaminated salad ingredients, raw seafood, infected food workers | Use potable water, practice good personal hygiene, cook all seafood |
12.8.2 Factors in Foodborne Disease
According to the World Health Organization (WHO), a small number of factors related to food handling are responsible for a large number of foodborne disease outbreaks worldwide. The most common factors and their reasons for contributing to foodborne disease are listed below:
| Factor | Effect |
|---|---|
| Preparation of food several hours in advance & improper storage | favors growth of pathogenic bacteria and/or formation of toxins |
| Insufficient cooking or reheating of food | not sufficient heat is applied to eliminate/reduce pathogens |
| Inadequate cooling of foods | permits survival of pathogens, allowing them to grow to sufficiently large numbers |
| Cross-contamination | allowing un-contaminated food to become contaminated by improper handling of food |
| Poor personal hygiene | can promote cross-contamination |
Why were foods implicated in foodborne disease outbreaks consumed, if they were so heavily contaminated with disease-causing microorganisms?
In some cases the food probably contained millions or billions of such bacteria per gram or millilitre, but the food would have appeared perfectly normal.
It is important to note that most pathogenic bacteria do not alter the colour, odour, flavour or texture of food even though they may be present in large numbers. This is the reason why perishable food must be handled in such a manner that contaminants will be destroyed during cooking or processing and that post-cooking and post-processing contamination must be avoided. In addition, perishable, low acid foods must be handled as though they were contaminated, which means that safe food handling practices must be followed at all times.
Which foods should we be particularly careful about?
Remember the principles we have learned in earlier lessons about factors affecting microbial growth and survival such as pH (acidity), moisture, time-temperature, oxygen.
Think about whether the food has been treated by some process that could decrease or inhibit microbes.
Consider possibility of post-processing contamination that may lead to an unsafe food.
For those who are at risk (e.g. the elderly, the very young, pregnant women and their unborn or newborn babies, and those who are immunocompromised) should be particularly careful about raw meat and fish, milk and eggs, and should avoid deli meats, soft cheeses, uncooked sprouts, liver pate, unpasteurized fruit juices/cider. They should take extra care in washing or peeling fresh fruits and vegetables, or may cook these before eating.
Top 4 places where food can be mishandled and lead to foodborne disease incidents:
- Food Service Establishments
- Food Processing Establishments
- Retail Food Establishments
- Homes
Clearly, the food service sector is responsible for the majority of reported foodborne disease incidents and cases. One of the problems in the food service sector is that foods are handled in large quantities. If proper handling procedures are not followed, it is easy for food to become contaminated, for cross contamination to occur, for inadequate cooling to occur, and for inadequate cooking or hot holding temperatures to occur since the food often receives much handling by many people before it reaches the consumer.
| Want to learn more? |
|
Although the food service sector may be responsible for many incidences of foodborne disease, such incidents were in fact attributed to all sectors of the food industry as well as in homes. It is the responsibility of the food processing industry and the regulatory agencies to ensure that changes in food technology do not lead to undue risks with respect to foodborne illness or other potential hazards. It may be worthwhile, at this point to consider how food is handled in your home.
What can be done to ensure food safety?
The World Health Organization (WHO) and the BC Health Files have developed ten rules for safe food handling, that if followed, can reduce the risk of foodborne disease.
Below are some key points that need to be considered
- Choose foods processed for safety (i.e. pasteurized products over unpasteurized)
- Cook foods thoroughly
- Eat cooked foods immediately
- Store food promptly and carefully. Perishable and "cold" foods should be kept "cold" below 4°C until ready to cook or eat.
- Avoid the "Temperature Danger Zone" (TDZ) which is from 4°C to 60°C (40-140°F). Bacteria can grow and/or produce toxins in food if left in the TDZ.
- Reheat cooked foods thoroughly
- Avoid cross-contamination. Do not allow any contact between raw food or its traces and cooked food
- Wash hands repeatedly
- Keep all kitchen surfaces clean
- Do not let anyone with diarrhea or infected sores prepare food
- Wash all fruits and vegetables before eating
- Finally, if in doubt, throw it out! (Ministry of Health Services, 2001)
12.8.3 The HACCP System: Seven steps to food safety
In the past, food safety programs run by the industry (food processors) and food service establishments, have corrected hazardous conditions after they happened. The HACCP system is designed to anticipate and control problems before they happen. The HACCP systems stands for Hazard Analysis and Critical Control Points. This system is recommended as the best method for ensuring food safety in retail establishments and food processing operations. There are 7 principles in a HACCP system:
- Identify Hazards
- Determine the critical control points (CCPs)
- Establish control measures (critical limits or thres holds)
- Establish procedures to monitor CCPs
- Establish corrective actions
- Keep records
- Verify procedures
While each principle is unique, they all work together to form the basic structure of an effective food safety program. (Source: McSwane, D., Rue, N., Linton, r., and Reeves, D.2003. Essentials of Food Safety and Sanitation. Pearson Education Canada Inc., p. 104-121)
HACCP Video
The food safety rules described above and the description of the seven steps for HACCP are summarized in the following video:
HACCP: The Hazard Analysis and Critical Control Point System
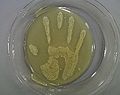
How important is that we wash our hands?
The following figure dramatically illustrates the importance of washing hands thoroughly after handling raw meats. In this experiment, a previously sanitized hand was rubbed with a cube of raw pork for about 15 seconds. The hand was then pressed gently onto a 9" agar plate and labeled as "soiled hand" (A). Hands were again washed with Hibitane hand sanitizer (4% w/v chlorhexidene gluconate) for 30 seconds and the right hand was gently pressed onto a second agar plate labeled as "washed hand" (B). Both plates were incubated at room temperature for 2 days. The images below show the results/observations after two days:
-
A. Soiled Hand
-
B. Washed Hand
The importance of washing hands. Right palm soiled with raw pork cubes (photograph courtesy of Eunice Yao, 2000. "Digital Image Analysis of Full-hand Touch Plates: A method of mimicking real soil encountered in food science industry to analyze the efficacy of antimicrobial hand sanitizer and alcohol-based sanitizing gels").
Microbe Video Pt.1
Microbe Video Pt.2
Microbe Video Pt.3
12.9 Summary of Lesson 12
This lesson has been a brief overview of the topic of toxicants in foods. There are hundreds of toxicants that could be found in our food supply. We are very fortunate that here in Canada, we have the food inspection systems and regulatory mechanisms that help insure that levels of toxicants in foods are kept at levels below the no-effect level.
In this lesson, you have also learned about the causes of foodborne illness in Canada. You have gained some insight into the importance of different types of microorganisms involved in foodborne disease incidents. You have learned the importance of considering different factors in assessing the hazardous nature of toxicants or foodborne illness caused by microorganisms
Through the assigned readings, you should also have become familiar with safe food handling practices, which you should employ whenever you handle food (clean, cook, separate and chill!)
You are introduced to the concept of HACCP which is a system designed to anticipate and control food safety problems before they happen. This is accomplished with the 7 steps of the HACCP system.
Remember that the risks of food poisoning and foodborne illness are dependent on the toxicant or pathogen itself, the susceptibility of the host and factors related to the environment and handling practices.
The concepts in this lesson will help you to apply what you have learned in your daily life to minimize the risks for yourself, your family and friends.
Supplemental Videos:
References
Authorship:
FNH 200 Course content on this wiki page and associated lesson pages was originally authored by Drs. Brent Skura, Andrea Liceaga, and Eunice Li-Chan. Ongoing edits and updates are contributed by past and current instructors including Drs. Andrea Liceaga, Azita Madadi-Noei, Nooshin Alizadeh-Pasdar, and Judy Chan.
|
|