Documentation:CHBE Exam Wiki/6.5 - Heat of Reaction and Formation Methods
6.5 – Heat of Reaction and Formation Methods
6.5.0 – Learning Objectives
By the end of this section you should be able to:
- Understand when you should use heat of reaction method.
- Understand when you should use heat of formation method.
- Solve a problem using heat of reaction method.
- Solve a problem using heat of formation method.
6.5.1 – Introduction
There are two common methods to solving energy balances. The first is heat of reaction method. This method is generally preferred for a single reaction sys where is known. The second method is heat of formation method. This method is generally preferred for multiple reactions and systems where is not readily available.
6.5.2 – Heat of Reaction Method
There are seven steps to solving an energy balance using the heat of reaction method:
- Solve the material balances as much as possible.
- Choose reference states for energy calculations.
- Calculate the extent of reaction for all reactions.
- Prepare the inlet/outlet enthalpy table.
- Calculate Enthalpies.
- Calculate for the reactor.
- Solve the energy balance.
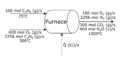
Let's try using this method to solve an energy balance. Suppose you are trying to solve how much heat is given off by the furnace shown below?
-
Attribution: Said Zaid-Alkailani & UBC
1. Solve the material balances as much as possible:
In this problem, the material balance has already been solved
2. Choose reference states for energy calculations:
For the reacting species, , , , , we will choose and atm as our reference state so that we have the same reference as the heat of reaction.
For the non-reacting species, , we will choose and atm as our reference state because we have an enthalpy table.
3. Calculate the extent of reaction for all reactions:
In this case we have 1 reaction and can use the formula (for any compound involved in the reaction):
4. Prepare the inlet/outlet enthalpy table:
-
Attribution: Said Zaid-Alkailani & UBC
5. Calculate Enthalpies:
-
Attribution: Said Zaid-Alkailani & UBC
-
Attribution: Said Zaid-Alkailani & UBC
6. Calculate for the reactor:
7. Solve the energy balance:
where , , and are assumed to be negligible.
6.5.3 – Heat of Formation Method
There are six steps to solving an energy balance using the heat of formation method:
- Solve the material balances as much as possible.
- Choose reference states for energy calculations.
- Prepare the inlet/outlet enthalpy table.
- Calculate Enthalpies.
- Calculate for the reactor.
- Solve the energy balance.
Let's try using this method to solve an energy balance. Suppose you are trying to solve how much heat is given off by the furnace shown below.
-
Attribution: Said Zaid-Alkailani & UBC
1. Solve the material balances as much as possible:
In this problem, the material balance has already been solved
2. Choose reference states for energy calculations:
For the reacting species, we must find their elemental species at standard condition. The elemental species for , , , and are , and . We will choose and atm as our reference state for these elemental species.
For the non-reacting species, , we will choose and atm as our reference state because we have an enthalpy table.
3. Prepare the inlet/outlet enthalpy table:
-
Attribution: Said Zaid-Alkailani & UBC
4. Calculate Enthalpies:
-
Attribution: Said Zaid-Alkailani & UBC
-
Attribution: Said Zaid-Alkailani & UBC
=== 5. Calculate
6. Solve the energy balance:
where , , and are assumed to be negligible.