Documentation:CHBE Exam Wiki/1.11 - Practice Problem 1
1.11 – Practice Problem 1
Question
In wastewater treatment, an important step in the process is sedimentation. Sedimentation is the process of allowing particulate matter in a fluid to settle to the bottom of a tank. In the sedimentation step, a coagulant can be used to coagulate bacteria and fine particles to allow the sedimentation to occur faster. The most common molecular species used for coagulation is alum, .
You are a chemical engineer working in a wastewater treatment plant. You are asked to make a material balance on the sedimentation process. Wastewater flows into a sedimentation tank at 200 kg/s. The wastewater contains 0.01 wt% of bacteria, 2 wt% particulate matter, and the balance pure water. Pure alum is added to the sedimentation tank in a separate stream. Alum allows 80% of the bacteria and 100 % of the particulate matter to settle. Just enough alum is added for this ratio of settlement. The sludge at the bottom of the tank exits the tank with 20 wt% of water and 40 wt% of particulate matter. The semi-treated water exits the tank in a separate stream.
a) Draw and label a block flow diagram (BFD).
b) Perform a degree of freedom analysis.
c) Fully balance the remainder of the block flow diagram.
Answer
a) Draw and label a block flow diagram (BFD)
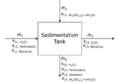
First, let's draw the basic layout of the BFD:
-
Attribution: Said Zaid-Alkailani & UBC
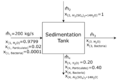
Using the information given, let's fill out as much of the BFD as we can.
-
Attribution: Said Zaid-Alkailani & UBC
b) Perform a degree of freedom analysis
We have 7 unknowns, so we need to find 7 equations. First lets do all of the mass balances:
Now lets do the mass fraction balance:
Next lets write our the extra equation:
Finally we can solve for the degrees of freedom:
c) Fully balance the remainder of the block flow diagram
To fully we balance the diagram we must first solve for all of the unknowns.
1. Solve for using particulate balance:
2. Solve for using the extra equation:
==== 3. Solve for using the fractional mass balance:
4. Solve for using the alum balance:
5. Solve for using a combination of the water balance and bacteria balance:
We can equate these two equations to get:
and using the fractional mass balance
we can solve for
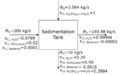
Now that we have completely solved the material balance, we can complete our BFD:
-
Attribution: Said Zaid-Alkailani & UBC