Course:FNH200/Lessons/Lesson 03
Fat and Sugar Substitutes - Sensory Perception of Foods
3.0 Overview
In this lesson, you will learn about ingredients that may be used to replace or substitute for fat and sugar components in foods. You will also learn about the factors that affect our perception of food quality, from the perspective of appearance, flavour and texture. We will examine the basic tastes and the food constituents that elicit those tastes. Flavour enhancers will be briefly covered in terms of what they are, how they function, and why they are used in foods.
Objectives
After completing this lesson, you will be able to:
- compare and contrast the different types of fat replacers and alternative sweeteners used in foods.
- discuss the role that sensory parameters play in our selection and perceptions of food
Optional Readings
- Canadian Diabetes Association, 2018. Sugars and Sweeteners https://www.diabetes.ca/DiabetesCanadaWebsite/media/Managing-My-Diabetes/Tools%20and%20Resources/sugars-and-sweeteners.pdf?ext=.pdf
- Fat Replacers, IFT scientific summary.1998. http://www.ift.org/knowledge-center/read-ift-publications/science-reports/scientific-status-summaries/fat-replacers.aspx
- Kroger, M, Meister, K. and Kava, R. 2006. Low-calorie sweeteners and other sugar substitutes: A review of the safety issues. Comprehensive Reviews in Food Science and Food Safety 5: 35-47.
- Réjeanne Gougeon, Mark Spidel, Kristy Lee, Catherine J. Field. 2004. Canadian Diabetes Association National Nutrition Committee technical review: Non-nutritive. Canadian J. Diabetes, 28(4):385-399. http://www.everstevia.com/CandianDiabetesAsso.pdf
3.1 Fat and Sugar Substitutes
Terms to remember |
|
Why use fat and sugar substitutes?
As you will recall from Lesson 1, one major consumer demand is for foods that contain less fat and sugar, and are overall healthier.
- Excessive caloric consumption along with a sedentary lifestyle are risk factors for obesity and other health problems.
- The food industry has responded to consumer demands by offering an ever-increasing variety of food products or ingredients that are low-fat or low-calorie, without sacrificing the taste and texture that consumers demand.
- Most of this research is taking place in developed countries where problems related to overeating and excess body weight are of major concern.
- Some sweeteners have been developed to allow consumers to enjoy sweet products without an excessive caloric intake
- Some sweeteners are non- or even anti-cariogenic (do not promote tooth decay)
- Some sweeteners are acceptable for diabetics based on their glycemic response rating
| Want to learn more? |
|
3.1.1 Types of Fat Substitutes
Fat substitutes are grouped as either protein, carbohydrate or fat based. The following table has an example of each of the 3 main groups:
| Type of Fat substitute | |
|---|---|
| Protein based: use protein particles to stabilize and give texture to food. Usually digested as protein | |
| Simplesse ® | Based on soy, milk or egg white protein.
The protein is partially coagulated by heat, creating a micro dispersion, in a process known as microparticulation. The spheroidal particles in this dispersion are very small (0.01-3 microns)! Due to the small particle size of the protein, we perceive the dispersion as a fluid with similar creaminess and richness of fat. Simplesse is digested as a protein, but due to the micro dispersion formed, produces only 1.0-1.3 Cal/g Applications include: Ice cream, yogurt, cheese spread, salad dressings, margarine, mayonnaise, coffee creamer, soups and sauces. |
| Carbohydrate based: They imitate fat's mouthfeel while contributing to less calories. Include cellulose, gums, modified starches, etc. | |
| Maltodextrins
Also known as Maltrin ® |
Derived from carbohydrate sources such as corn, potato, wheat and tapioca. It produces a smooth mouthfeel and bland flavour
Maltrin is fully digestible, yielding 4 Cal/g (remember that fat yields 9 Cal/g). Other carbohydrate based fat replacers are available that range from non-digestible to partially digestible (0-2 Cal/g) Applications include: Margarine, salad dressings, frozen desserts, frostings, processed meat. |
| Fat based: Some are made form long and/or short fatty acid chains. Others have fatty acids linked to sucrose (instead of glycerol- as in normal fat) | |
| Olestra
Also known as Olean ® |
Made from a sucrose molecule and 6-8 long-chain edible fatty acids forming a sucrose polyester.
Unlike other fat substitutes, Olestra can withstand high temperatures (e.g. frying), and gives the rich taste and creamy texture of characteristic of fat because it is made primarily from fat. Enzymes that breakdown ordinary fat can not break down Olestra, therefore it passes through the body ‘unchanged’ (Olestra is not metabolized and not absorbed by the human body), contributing to 0 Cal/g. Products containing Olestra must mention that vitamins A, D, E and K have been added (see below) Olestra was approved in the U.S.A. in 1996. Currently it is not approved for use in Canada. Applications include: Salty, savory snacks and crackers |
"Simplesse" and "Maltrin" are trademarked or registered brand names, therefore you may not necessarily see these names on the label of a food product. Look instead for the proteins and carbohydrates listed as ingredients.
| Want to learn more? |
|
3.1.2 Types of Sugar Substitutes - Sweeteners
In Canada, Section B.01.001, of the Food and Drug Regulations (FDR), defines sweetener as "any food additive listed as a sweetener in Table IX to B.16.100." Examples of sweeteners are: aspartame, sucralose, sorbitol, and maltitol.
In contrast, the FDR defines Sweetening agent as "any food for which a standard is provided in Division 18 of the FDR, but does not include those food additives listed in the table to Division 16 [B.01.001]". Examples of sweetening agents are: sugar, honey and molasses.
Definitions of "sweeteners" versus "sweetening agents" are given at:
For information on "FOOD ADDITIVES THAT MAY BE USED AS SWEETENERS" in Canada:
( scroll down to Division 16 and look at Table IX )
As we mentioned at the beginning of this lesson, sweeteners may be preferred or necessary for: Individuals with diabetes, those concerned with high caloric intake, and consumers trying to reduce the risk of tooth decay (cavities).
Sweeteners may be naturally occurring or synthetic molecules. Many of them were discovered serendipitously! For example, naturally occurring sweet peptides and proteins, derivatives of sugars (mono, disaccharides) and derivatives of amino acids or small peptides.
Low-calorie sweeteners

Aspartame is a methyl ester of a dipeptide composed of 2 amino acids (phenylalanine and aspartic acid). It is metabolized as proteins (amino acids), contributing to 4 Cal/g.
Aspartame is 180-220 times sweeter than sucrose, which allows for very small amounts to be used (thus, a low-calorie sweetener). It also does not increase blood glucose or insulin levels. However, people suffering from the rare metabolic disorder known as "PKU" (phenylketonuria) must avoid aspartame.
Aspartame undergoes degradation reactions at high temperatures (can not be used on baked goods), and eventually degrades overtime into DKP (diketopiperazine). A best before date is necessary on products with aspartame.
Applications include: Acidified beverages, desserts, frozen products, breakfast cereals.
Sugar-alcohols: maltitol, sorbitol, mannitol, isomalt, xylitol
Found naturally in a wide variety of fruits and berries, and also commercially produced by hydrogenating sugars.
Mainly used as a "Bulk sweeteners" (can be used cup-for-cup [volume-for-volume] in the same amount as sugar). They are also responsible for the cool-refreshing (menthol-like) sensation perceived from products like chewing gum and toothpaste,
Unlike the other sweeteners, sugar alcohols are less sweet than sugar (e.g. sorbitol is 60% as sweet as sucrose).
Do not promote tooth decay as they are not fermentable by the bacteria in our mouth.
Sugar alcohols also do not increase blood glucose or insulin levels. They are absorbed slowly in the large intestine, thus contributing 1.5-3.0 Cal/g. This 'slow absorption' can lead to a laxative effect when excess consumption occurs.
Applications include: Chewing gums, candies, frozen desserts, cookies, cakes, icings and fillings as well as oral care products(including toothpaste and mouthwash).
Non-caloric sweeteners
Acesulfame-k can provide a synergistic sweetening effect when combined with other sweeteners (e.g. used with Aspartame in soft drinks).
It is heat stable. It has a high degree of stability over a wide range of pH and temperature storage conditions.
Acesulfame-K is 200 times sweeter than sucrose and not metabolized by the body (contributes to 0 Cal/g). It is not fermented by oral bacteria, so it does not contribute to the development of cavities
Applications include: Baked goods, candies, canned goods, chewing gum dry foods. Also used in oral hygiene and pharmaceutical products
Sucralose is a chlorinated molecule in which 3 hydroxyl groups(OH) of the sucrose molecule are replaced by chlorine. It tastes similar to sugar but it is 600 times sweeter than sucrose. Sucralose is not metabolized by the human body, passing through unchanged (contributes to 0 Cal/g).
Sucralose is heat stable. It retains its sweetness over a wide range of temperature and storage conditions and in solutions over time. It has no effect in carbohydrate metabolism and does not increase blood glucose or insulin levels. It also does not support the growth of oral bacteria (does not promote tooth decay).
Applications include: Canned fruit, fruit drinks, baked goods, chewing gum, tabletop sweeteners, maple syrup, apple sauce.
Steviol glycoside is derived from the leaves of the South American Stevia plant where it has been used by indigenous people for centuries. The steviol glycosides are 100-150 times sweeter than sucrose and provide 0 Cal (due to the intense sweetness), and do not increase blood glucose or insulin levels and does not promote tooth decay. Stevia also remains stable under acidic conditions and high temperature does not destroy its sweetening properties.
| Want to find out more? |
|
The table below lists the acceptable daily intake (ADI) for several sweeteners. Further details in how the ADI is determined will be covered in Lesson 4: Food Additiives.
| Approved Sweeteners in Canada | Acceptable Daily Intake2 (mg/kg body weight) |
| Acesulfame-K | 15 |
| Advantame | 5 |
| Aspartame | 40 |
| Erythritol | 1000 |
| Neotame | 2.0 |
| Polydextrose | |
| Sucralose | 8.8-9.0 |
| Stevia extracts (steviol glycoside) | 2.0-5.0 |
| Sugar alcohols | unspecified, GMP1 |
| Saccharin | 5.0 |
1 Good Manufacturing Practice: minimum amount of an additive required to accomplish the specific purpose for which the additive is listed.
2 Daily dosage of a chemical which during an entire lifetime appears to be without appreciable risk on the basis of all facts known at that time. The acceptable daily intake is expressed as mg intake per kg body weight.
3.2 Sensory Properties of Foods
Sensory evaluation is defined as a scientific discipline used to evoke, measure, analyze and interpret reactions to those characteristics of foods and materials as they are perceived by the senses of sight, smell, taste, touch and hearing.
Sensory evaluation of foods is widely used in the food industry as an important tool for: new product development, product matching, shelf-life studies, product reformulation, quality control, and consumer preference, among others.
What makes us decide on the type of food we consume?
Our decisions to consume a particular food item or combination of food items are based in part on nutritional factors but for the most part are often driven by sensory characteristics of the foods.
In order to better understand the concept of sensory properties of food, you should do the following:
- Make a selection of a food item in the grocery store or at your dinner table, consciously thinking about your approach to food selection.
- What are your basic criteria for accepting a particular food item?
- Follow the guidelines below to help you.
- Our first step in selecting a particular item is based upon appearance of the food (colour, shape, size, gloss). For example, if a new variety of orange were offered for sale we might reject that orange if it was red instead of orange in colour. We will also reject a strawberry that was green instead of red, thinking that it is not ripe.
- If the appearance of the food item is acceptable then the next step in the selection process is to determine the odour of the food through our sense of smell. For example, the aroma of fresh baked bread and cooked bacon.
- If that is acceptable we then examine the texture of the food through the sense of touch. For example, fresh bread has a soft texture, the crispiness in a potato chip, the thickness of maple syrup.
- Finally we decide whether to put the food item in our mouth in order to determine the flavour and texture in the mouth.
If any of the previous parameters noted above fall outside of our preset limits of acceptable quality for that particular food item we will reject that food item. The interrelationships among various sensory factors are shown in Figure 3.1.

Appearance Factors
Our perception of the appearance of a food is governed by a number of factors such as size, shape, colour, gloss, consistency, and presence of defects (e.g. mould on an orange; bruises on an apple).
When you watch television or read a magazine next time, pay particular attention to the food advertisements and determine how the advertising appeals to your senses. Advertisements describing the snap, crackle and pop of a breakfast cereal, the crunch of a particular brand of pickle, the thick consistency of a particular brand of catsup or the refreshing, clean flavour of a particular brand of soft drink are designed to appeal to the senses we use in making food selections.
Appearance factors are used as a measure of food quality. Thus mould growing on an orange is indicative of a spoiled product. Bruises on an apple or peach are indicative of poor handling and storage. The bright, shiny red and green of a freshly picked apple are indicative of freshness and superior quality.
This is one of the reasons why appearance of food products, whether produced by a food processor, a restaurateur or by yourself in your home receive a great deal of attention.
Textural Factors
Textural parameters are often used in food selection and in food quality measurement. Four examples of textural parameters are shown in Figure 3.2.
When you select bread and buns by gently squeezing them you are applying a measurement of texture, that is the resistance of the bread to deformation under an applied force and also the ability of that bread to regain its shape after the force is released. The fresher the bread the less force required to deform it while an older loaf of bread will appear "tougher" because the starch has begun to undergo retrogradation (Lesson 2) and thus the bread does not appear to be as soft and fresh.
We sense the toughness of meat when we cut it with a knife and with our teeth and facial muscles when we chew it. The more force required to cut the meat, the less tender the cut and our perception is that of the meat is of lower quality.
| Want to learn more? |
|
Flavour Factors
Flavour is a sensation made up of a combination of two senses: FLAVOUR = TASTE + AROMA (SMELL). In order to perceive a full bodied flavour, we need both the perception of TASTE and AROMA.
To elicit a taste sensation, a substance must be water-soluble and it must interact with the appropriate sensory receptors on the tongue.
Taste is detected in the mouth and primarily on the tongue.
To elicit aroma, substances must be fat soluble and volatile in order for them to interact with the odour or aroma receptors in the olfactory region of our nasal passages. Aromas of foods are a complex mixture of chemicals, which are often present in foods in very low concentration. Aroma constituents are a very important part of foods in relation to our perception of food quality.
When we have a cold, why does food seem bland?
When you have a cold and your sinuses are congested, food often has a bland flavour, basically being a combination of saltiness, sourness, sweetness and bitterness, which are the basic tastes.
Because your sinuses are congested you do not detect odour or only detect a very small amount of odour when consuming the food and thus it appears to be bland.
Description of the taste receptors
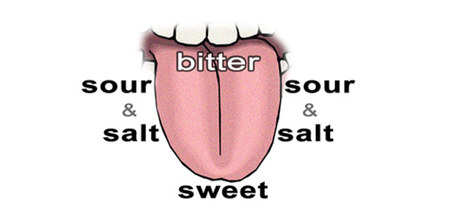
There are four recognized taste sensations: sweet, salty, sour and bitter. Some researchers recognize a fifth taste, called "umami". A diagram of the tongue with the locations of the four basic types of taste receptors is shown in Figure 3.3 - in fact, more recent research suggests that the four types of taste receptors are not as localized as shown in this figure, although they are more predominant in those locations.
Take a look at the artistic rendition of a taste bud in the article "Anatomy of Taste" published in Popular Science; Nov2007, Vol. 271 Issue 5, p48-49. Are you a "supertaster"?
Sweet
Sweet sensations are elicited by simple sugars, especially monosaccharides; the sweetness of oligosaccharides decreases as the number of sugar residues increases. Sugars are not the only sweet compounds. Some amino acids such as glycine are sweet. Some peptides (composed of two or more amino acids) are sweet; a notable example is Aspartame.
Aspartame, the low-calorie sweetener discussed earlier in this Lesson, is a dipeptide made of aspartic acid and phenylalanine, two amino acids commonly found in proteins in foods.
Other sweeteners include the synthetic compounds such as cyclamate and saccharin, which are neither amino acids nor carbohydrates. There are other compounds such as chloroform and lead acetate, which are sweet but not used as sweeteners!
Salty
Only one compound, namely sodium chloride, produces a pure salty taste. Compounds such as potassium chloride, often an ingredient in salt substitutes, produces a salty as well as a bitter taste. This is one of the reasons why it has been very difficult to formulate a palatable salt substitute for individuals on low sodium diets. It is not the sodium portion or the chloride portion of sodium chloride that elicits the salty taste. It is the ionized molecule of sodium chloride that it required for the production of a salty taste. Thus sodium sulphate is bitter but only slightly salty while calcium chloride is very bitter and cesium chloride is sweet.
Sour
Sour tastes are produced by protonated organic and inorganic acids. Citric, tartaric, malic, lactic, fumaric, acetic and phosphoric acids produce a sour taste and are commonly found naturally in many acidic foods and are also used as acidulants in foods. The acid taste of vinegar is due to acetic acid. You can probably recall many instances where you use vinegar to impart an acid taste to a food.
Bitter
Compounds that are bitter are typically alkaloids such as caffeine (in coffee and tea), theobromine (in chocolate) and solanine (a naturally occurring toxicant in green potatoes). Some salts such as sodium sulphate and calcium chloride are bitter, as are some amino acids and peptides. "Bitter peptides" contribute to the sharpness and bitterness of aged Cheddar cheese.
Umami
Umami is described as a "savoury" and "delicious" sensation, and is associated with compounds known as "Flavour Enhancers or Potentiators". Flavour enhancers are compounds that elicit no taste of their own at low concentrations, but can modify the perceived intensity or quality of the taste produced by another substance.
A typical example is monosodium glutamate, more commonly known as MSG. It apparently binds to the taste receptors in the tongue and causes an enhancement of taste sensations. Other flavour enhancers are the 5'-nucleotides such as inosine 5'-monophosphate, which enhances meaty flavour.
MSG has no effect on the aroma of a food. It enhances meat and vegetable flavours but does not enhance the flavour of acidic foods such as fruit, bakery products or sweet products. Enhancement of meaty flavours occurs at MSG concentrations below the level where MSG itself produces a typical taste sensation. MSG suppresses hydrolysed vegetable flavours (bitter) and sulfur or burnt cabbage flavour notes in foods.
| Want to learn more? |
|
Maltol, is also considered as a flavour enhancer. It modifies the flavours of soft drinks, fruit drinks, jams and other high carbohydrate foods. However, it does not elicit an "umami" (savory) sensation!
Other sensations perceived in the mouth/tongue:
Some specific chemical components of foods may lead to warming, cooling or other sensations in the mouth, tongue and lips.
- Astringency is more of a "physical" sensation described as puckering in the mouth; it is most often attributed to tannins or polyphenols of high molecular weight.
- Pungency is the term used to describe the sensation of "spicy heat" in the oral cavity. A well known example of pungent substances is the capsaicinoid family of molecules, such as capsaicin, found in chili peppers.
- In contrast, the sensation of coolness is a familiar one to you if you like to chew gum. Key compounds responsible for the cooling effect are menthol and its isomers. Various sugar alcohols such as xylitol and sorbitol also produce a cooling effect.
From the preceding discussion you should have some appreciation of the fact that sensory parameters of foods are very complex.
Our perception of the sensory properties of foods are dependent not only on the chemical and physical properties of the food but also on psychological factors as well.
3.3 Sensory Science: Sensory Evaluation of Foods

Sensory evaluation is a scientific discipline using human subjects to evaluate food based on the sensation perceived by senses of sight, smell, taste, touch and hearing. Trained and untrained panellists can be used and simple to complex statistical analysis will be used to interpret the results. Systematic and scientific sensory evaluation was initiated during the world wide war to provide palatable food for troops and later developed into more advanced and scientific discipline and serving the economic interests of companies.
Sensory evaluation of foods is widely used in the food industry as an important tool for: new product development, product matching, shelf-life studies, product reformulation, quality control, and consumer preference, among others.
There are 3 main types of sensory tests (methods) used (Figure 3.4): Discriminative and Descriptive analyses (both of which are product-oriented and analytical), and the Hedonic/Preference/Acceptability tests (which are people-oriented and considered as "affective").

3.4 Discriminative or Difference Tests

Discrimination or “Difference” tests are used to determine whether a difference exists between samples. The difference can be defined or it may not. These tests would be used to evaluate if a new processing treatment, yeast type or aging treatment has changed the character of the food product. Panelists’ personal likes and dislikes are not a concern.
Difference tests are the backbone of sensory analyses. They allow the experimenter to document the presence of perceived differences among samples for quality control, product development and/or research and development purposes.
Examples of commonly used different tests include: triangle tests, pair-difference tests. Figure 3.5. shows an example of a triangle test set-up.
Descriptive Analysis Tests
Descriptive analysis requires detection, description and quantization of the sensory aspects of a product. It is used only with trained panelists. These trained panelists must be trained for several weeks in order to be able to be "calibrated" and accurately detect, describe and rate the intensity of each attribute (whether it is appearance, aroma, flavour, mouth feel, etc.). Examples of descriptive analysis tests include: Flavour profile method, quantitative descriptive analysis (QDATM), and free-choice profiling.
Hedonic/Preference Tests
Preference tests are also known as consumer tests. The objective of these tests is to evaluate a personal (subjective) response to a product. Consumers can give their preference between products, degree of liking of a product, or their overall acceptance of a product. Preference tests require a large number of panelists (100s to 1000s) in order to represent the target population for the product being tested. Examples of preference tests include: Paired-preference, ranking and hedonic scales.
3.5 Summary
- Fat substitutes can be made form proteins, carbohydrates or even some types/forms of fats! Different fat substitutes provide food with unique characteristics in terms of mouthfeel, texture, calories, etc.
- Sugar substitutes are also referred to by the public as "Artificial Sweeteners".
- Sweeteners vary in composition, sweetness level, calories, and applications.
- There are three main sensory factors that we use when evaluating food: Appearance, texture and flavour (includes aroma and taste)
- Sensory parameters of foods are very complex. Our perception of the sensory properties of foods are dependent not only on the chemical and physical properties of the food but also on psychological factors as well.
- Sensory Evaluation of foods is a scientific discipline widely used to measure and quantify our perceptions to food.
Authorship:
FNH 200 Course content on this wiki page and associated lesson pages was originally authored by Drs. Brent Skura, Andrea Liceaga, and Eunice Li-Chan. Ongoing edits and updates are contributed by past and current instructors including Drs. Andrea Liceaga, Azita Madadi-Noei, Nooshin Alizadeh-Pasdar, and Judy Chan.
|
|
