File:1-s2.0-S0092867402011364-gr7.jpg
1-s2.0-S0092867402011364-gr7.jpg (565 × 599 pixels, file size: 111 KB, MIME type: image/jpeg)
Summary
| Description | English: The Recognition Complex of Internalin and E-Cadherin in its Cellular Context
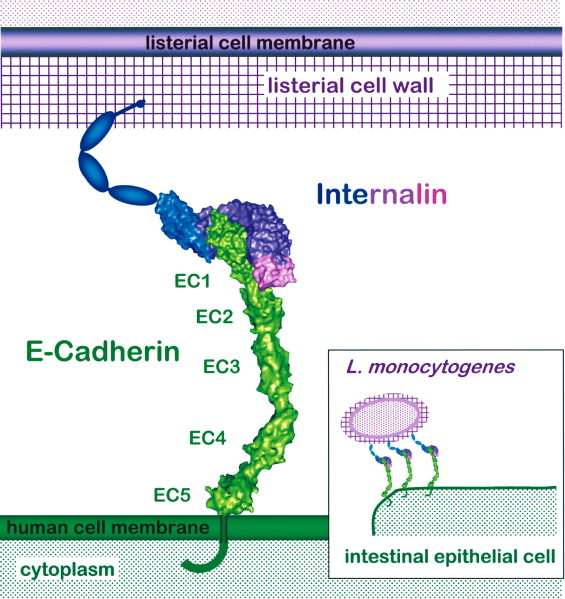
InlA, covalently bound to the cell wall of L. monocytogenes recognizes and binds E-cadherin. Known protein structures are represented by molecular surfaces: hEC2-hEC5 are modeled on C-cadherin (Boggon et al., 2002). C-terminal regions of both molecules are depicted schematically only. Inset: Both internalin and E-cadherin are present in high copy numbers on the surface of their respective cells. Schubert, W. D., Urbanke, C., Ziehm, T., Beier, V., Machner, M. P., Domann, E., ... & Heinz, D. W. (2002). Structure of internalin, a major invasion protein of Listeria monocytogenes, in complex with its human receptor E-cadherin. Cell, 111(6), 825-836. |
| Date | 12 February 2015 () |
| File source | http://www.sciencedirect.com/science/article/pii/S0092867402011364 |
| Author | Schubert, W. D., Urbanke, C., Ziehm, T., Beier, V., Machner, M. P., Domann, E., ... & Heinz, D. W. (2002). Structure of internalin, a major invasion protein of Listeria monocytogenes, in complex with its human receptor E-cadherin. Cell, 111(6), 825-836. |
Licensing
{{subst:uwl}}
File history
Click on a date/time to view the file as it appeared at that time.
| Date/Time | Thumbnail | Dimensions | User | Comment | |
|---|---|---|---|---|---|
| current | 07:56, 13 February 2015 |  | 565 × 599 (111 KB) | WaldenCheung (talk | contribs) | User created page with UploadWizard |
You cannot overwrite this file.
File usage
There are no pages that use this file.